This article talks about methods to improve EMI and EMC in medical devices.
Electromagnetic compatibility (EMC) of a device means that it is compatible with its electromagnetic environment and does not emit electromagnetic waves to a level that causes Electromagnetic Interference (EMI) in other devices. An increase in emission of electromagnetic energy of a device beyond a limit can disturb its own operation or of an electronic device operating nearby. So, manufacturers must design their devices with adequate protection against possible interferences.
The difference between immunity and emission testing is that while immunity requirement is concerned with how susceptible a device is to electromagnetic energy emitted from surrounding devices, emission requirement is concerned with the amount of electromagnetic energy emitted from the device.
IEC 60601-1-2 is the EMI/EMC standard for medical electrical equipment and systems. Medical devices require complex analysis of EMI/EMC regulations since these devices are no longer used in just a hospital setting. With the increasing use of wearable medical technology, patients can be anywhere in the world in any environmental condition. EMC standards must comply for improved designs of medical devices.
Importance of EMI shielding
Due to the increase in demand for medical electronic devices, development in this sector is happening fast. Resolution level is continuously increasing to provide greater details and more accurate diagnosis. To ensure an enhanced level of detail, it is necessary to create an environment where EMC and EMI do not interfere with the operation of medical electronic systems. Imaging devices must provide reliable and noise-free data even in harsh environments, like around MRI or X-ray systems.
EMI can fail monitoring systems or life-support equipment. Patients and individuals who rely on medical devices depend on EMI shielding. So, it is necessary to make sure that the devices function properly at all times.
There is a need for automation of simple devices as well as advanced medical equipment, to monitor patients and perform diagnosis remotely. This increases the demand for communication technologies like Bluetooth, WLAN, GPS and other wireless links. This again increases EMI as all types of wireless technologies face challenges in coexisting in the same space. As clock speeds and densities increased in embedded electronics, need for the higher signal-to-noise ratio (SNR) drove the need for EMI shielding.
Factors that affect EMC of a medical device
Power disturbances can occur due to electromagnetic waves emitted by communication devices or electrostatic discharges (ESDs), which create a conductive current surge and localised radiated transient field. With the increase in the use of cellular phones, devices emitting electromagnetic waves have become a part of the environment.
EMI might occur if the following three factors are present: source of EMI, coupling path and receptor. Coupling path from the source to the receptor can either be an electric current, magnetic field or an electromagnetic field.
EMI source can be a natural-like lighting; it can also come from such devices as radios, computers, wireless networks, cellphones or any electric devices designed to transmit signals. Many factors are responsible for causing EMI in medical devices, including:
- Design to interact with magnets, allowing these to be re-programmed or monitored using magnets from outside the body
- Use of magnetically-susceptible materials such as implantable devices, MRI and so on
- Current and voltage generated on leads or loops due to electromagnetic fields
The flow of design to improve EMI/EMC is as follows:
- Product description
- Defining noise factors
- Studying interaction results
- Determining possible solutions
- Setting required for test environment
- Conducting tests
- Calculating SNR
- Determining factor significance and optimum set of parameter levels
- Verifying and validating design
- Developing a prototype
- Final testing
- Time-to-market
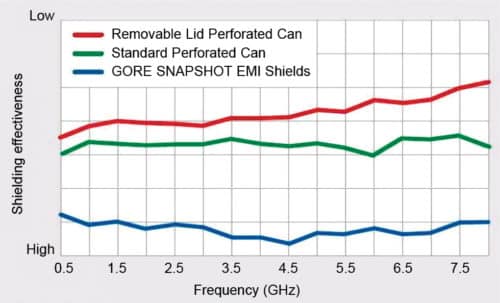
Multi-cavity EMI shields
Although the demand for more compact, lighter plastic equipment is on the rise, non-conductive plastic needs metal shielding to ensure protection from EMI. Medical device manufacturers and engineers consider EMI shielding to prevent medical equipment failure and keep these compliant with federal regulations.
A multi-cavity EMI shield solves many problems associated with shielding technologies. It consists of a lightweight metalised plastic material that can be moulded to fit any design. The plastic material is attached to the PCBs after SMT reflow process, allowing for easy component inspection or rework. It can be easily removed by hand without damaging the board or soldering. It has superior shielding capability and easy attachment mechanism as compared to perforated, soldered metal cans.
A multi-cavity EMI shield has great applications in broadband data centre routers, communication devices, handheld military radios, industrial barcode scanners and mobile consumer electronics. XGR Technologies is an EMI shielding solution provider, and SnapShot shield is its major product. It is available in flexible designs such as circle, square, parallelogram and more.
Bill Candy, chief technology officer, XGR Technologies, says, “Often times in medical devices, distance between different electrical operations needs to be minimised and fit into a specific form factor. With flexible design shapes available with SnapShot shielding technology, the device form factor need not be altered to fit a typical rectangular shield. SnapShot shield can conform to the shape necessary for the specific device.
“Additionally, for medical devices such as MRI machines that are sensitive to magnetic materials, using a polymer-based non-ferromagnetic shield such as SnapShot allows components to be placed closer to the imaging head, that is, within the magnetic field of the device. This leads to better image quality in these devices.”
EMC/EMI filters
An EMI filter can assist with conducting emissions and meeting immunity and fast transient requirements of radiated emissions. A power line or mains EMI filter is installed at the power entry point of the equipment to prevent electromagnetic noise from exiting or entering the equipment. This can protect military and aerospace equipment from failure due to EMI noise.
Design parameters for selecting an appropriate EMI filter include attenuation or insertion loss, load impedance, rated current, rated voltage and regulatory approval requirements specified by the user. However, there are many other parameters that must be considered to get the most reliable, efficient and proper operation from the filter.
Value of capacitors connected between lines to protective earth (PE) is directly related to leakage currents in the filter. As per medical standards, ground leakage current should be restricted in medical electrical equipment. Usually, an EMC/EMI filter does not have any capacitor to ground. This reduces the performance of the filter. As a result, a high-performance medical filter requires additional inductance to cancel out common mode noise.
Design of a custom filter solution depends on the level of insulation needed. The filter should not introduce any additional leakage current due to the parasitic effect in the system. However, an EMC filter with very low leakage current can be considered in less critical applications. Even with a filter that does not introduce any leakage current, overall leakage current for the whole system needs to be determined.
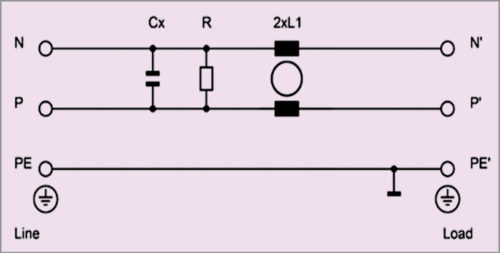
Schaffner Impulse provides FN92XX filters (B-type in particular) compliant with the requirements of EN/IEC 60601-1 standard regarding leakage current and voltage withstand. It offers standard solutions for different levels of leakage current requirements. Its B-type filters do not have any capacitance to PE and therefore do not add any leakage current to the system.
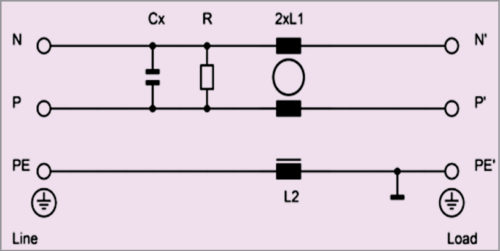
It also offers EB-type filter to compensate some of the lost performance as a result of no capacitance to PE. This type also increases immunity to electrical fast transients compared to B-type.
Electroplating to improve reliability and functionality of medical electronic devices
A metal coating protects against EMI without adding too much weight to medical equipment. It also strengthens digital equipment, extending its lifespan. Conductive materials, like copper, accomplish EMI protection by blocking electromagnetic emissions and then reflecting and absorbing them. An EMI shield can be made from a metal enclosure or metal coating. Using a conductive metal as a shield does not affect speed—it only blocks radio waves and other forms of EMI.
Electroplating can be used to coat parts with copper, nickel, gold, silver and other metals needed for EMI shielding. Sharretts Plating adheres to quality certifications such as ISO 13485 and ISO:9001 by providing required electroplating facilities. It makes sure that products are biocompatible by decreasing the risk of infection or rejection of implantable devices.
Conclusion
EMC filters for medical applications should fulfill the most stringent leakage current regulations, offer the best size versus performance and should be designed for typical usage in the medical environment.
EMI shielding is becoming more challenging because people are using medical devices also at homes, on the go and anywhere they need. According to Institute of Electrical and Electronics Engineers (IEEE), portable health devices tend to be more susceptible to EMI because these use low-power circuitry that can be more sensitive to electromagnetic fields. Medical devices are also becoming smaller and faster, and are being constructed with digital circuits. This poses as a challenge for device engineers because of faster digital speed and greater bandwidth requirement, making it more difficult to control electromagnetic emissions and their vulnerability.



