Researchers at Pohang University of Science and Technology have developed a new anode material for batteries, enabling electric vehicles to charge in just six minutes.
Charging an electric vehicle typically requires 10 hours or more, and even with fast-charging techniques, it still takes a minimum of 30 minutes, provided a charging spot is available. Accelerating the charging process to match the refuelling speed of gasoline-powered cars would address the scarcity of EV charging stations. The efficiency of lithium-ion batteries, commonly used in electric vehicles, depends on the anode material’s ability to store lithium ions.
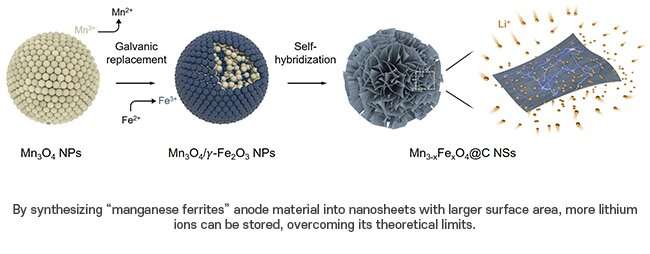
Pohang University of Science and Technology (POSTECH) researchers have developed a new anode material. The team successfully synthesised manganese ferrite nanosheets (Mn3-xFexO4) using an innovative self-hybridisation method involving a straightforward galvanic replacement-derived process. The technique increases storage capacity by 1.5x the theoretical limit, allowing electric vehicles to charge in just six minutes.
By combining manganese oxide with iron in a solution, a galvanic replacement reaction occurred, forming a heterostructure compound where manganese oxide was positioned internally, and iron oxide resided externally. Using a hydrothermal method, the team generated nanometer-thick sheets of manganese ferrites, which exhibited increased surface areas. Leveraging highly spin-polarised electrons, this technique significantly augmented the storage capacity, accommodating many lithium ions. This innovative approach surpassed the theoretical capacity of the manganese ferrite anode material by over 50 per cent.
Expanding the surface area of the anode material enabled enhanced movement of a substantial number of lithium ions, resulting in faster battery charging. Experimental findings demonstrated that a battery with a capacity equivalent to those utilised in current market EVs could be charged and discharged in as little as six minutes. The study has successfully refined the complex synthesis process, achieving a breakthrough in the theoretical capacity of the anode material while significantly accelerating the battery charging process.
The team provided fresh insights into overcoming electrochemical constraints in traditional anode materials and elevating battery capacity through strategic surface modifications utilising electron spin. The researchers believe this advancement could enhance battery longevity and decrease recharging duration in electric vehicles.
Reference: Song Kyu Kang et al, Exceeding Theoretical Capacity in Exfoliated Ultrathin Manganese Ferrite Nanosheets via Galvanic Replacement‐Derived Self‐Hybridization for Fast Rechargeable Lithium‐Ion Batteries, Advanced Functional Materials (2023). DOI: 10.1002/adfm.202300143






