Unlike conventional hydrogen production systems reliant on fossil fuels, this new system exclusively harnesses solar energy.
MIT engineers are revolutionising hydrogen production, eliminating fossil fuels. Their reactor system, resembling a train, solely relies on solar energy. A recent Solar Energy Journal article details their blueprint for “solar thermochemical hydrogen” production. This clean energy method, powered by the sun’s heat, directly splits water, generating hydrogen for eco-friendly long-distance transportation without greenhouse gas emissions.
Hydrogen production depends heavily on fossil fuels, tarnishing its green potential. Solar thermochemical hydrogen (STCH) offers a clean alternative, relying solely on renewable solar energy. Yet, current STCH designs are inefficient, utilising only 7% of incoming sunlight, leading to low yields and high costs. Aiming for solar-based fuels, the team envisions their design capturing 40% of solar thermal energy to boost hydrogen output. This efficiency cut costs, making STCH a scalable, economical solution for transportation decarbonisation.
Ahmed Ghoniem, the lead author and MIT’s Ronald C. Crane Professor of Mechanical Engineering, sees hydrogen as the future fuel. Their goal aligns with the Department of Energy’s objective: green hydrogen by 2030 at $1 per kilogram. Efficiency is critical to make this cost-effective, maximising solar energy use for hydrogen production.
Solar installations`
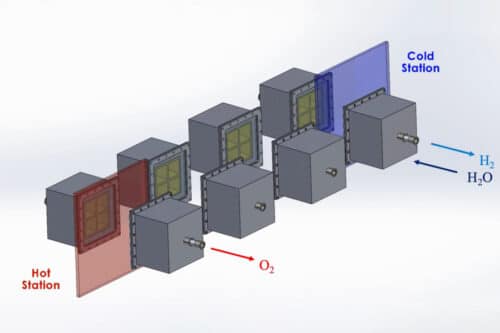
The proposed system, similar to other conceptual designs, integrates with an established solar heat source, such as a concentrated solar plant (CSP), characterised by a circular arrangement of mirrors directing sunlight toward a central tower. The STCH system captures the tower’s heat and channels it into the water-splitting process, contrasting with electrolysis, which uses electricity for water separation. At the heart of the STCH system lies a two-step thermochemical reaction. Initially, water vapour interacts with a metal, causing the metal to extract oxygen from the vapour, leaving behind hydrogen. This metal “oxidation” process resembles iron rusting in water but is significantly faster. After isolating hydrogen, the oxidised metal undergoes reheating in a vacuum, reversing rusting and revitalising the metal. Devoid of oxygen, the metal cools and re-engages with steam, enabling continuous hydrogen production, repeatable many times.
The system optimises this process with a circular track housing rectangular reactors encircling a solar thermal source, like a CSP tower. Each reactor is dedicated to the metal’s redox process. Reactors experience high heat in the sun (up to 1,500 degrees Celsius), extracting oxygen and leaving the metal “reduced.” They then transition to a more excellent station at around 1,000 degrees Celsius, where steam triggers hydrogen production.
Corrosion and Trackways
Efficiently managing cooling heat and reducing energy consumption in large-scale hydrogen production is addressed by using thermal radiation for heat exchange between reactors on a circular track. This design also eliminates the need for energy-intensive pumps by employing lower-temperature reactors to extract oxygen and reverse oxidation, ensuring continuous hydrogen and oxygen production.
The design significantly improves solar thermochemical hydrogen efficiency in simulations, from 7% in previous designs to an impressive 40%. Christopher Muhich, an Arizona State University chemical engineering professor, sees it as a potential game-changer for continuous hydrogen production, vital for converting sunlight into liquid fuels.
Over the next year, the team plans to build and test a prototype at the Department of Energy’s concentrated solar power facilities, their primary funding source. The vision is a modular system housed in a small structure amid a solar field, with multiple trains containing around 50 reactors. Additional reactors can be easily added for scalable hydrogen production.



