A light-based method uses light to check how fast blood clots, helping make implants safer, guide medicine, and even find tiny impurities in water.
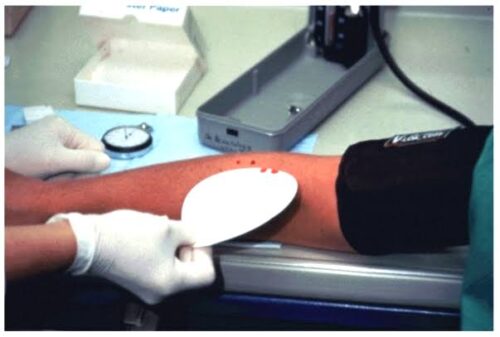
Researchers at the Indian Institute of Technology, Madras (IIT-M) have developed a patented method that measures blood clotting time with millisecond-level precision using changes in light reflectivity. The technique offers a highly accurate way to monitor how blood interacts with materials, helping prevent clot-related complications in medical implants and guiding precise dosing of anti-coagulation medication post-surgery.
The method works by detecting changes in the reflectivity of an implant’s surface. As blood begins to clot, the surface becomes cloudy, altering the reflected light, which is captured as a voltage shift by a photodetector. This provides quantitative data that is more precise than traditional methods, such as mechanical tilting or free haemoglobin tests, which are prone to inaccuracies.
By enabling accurate testing of materials for stents, heart valves, and catheters, the technique allows manufacturers to screen for haemocompatibility early in the research stage. Researchers also note that with minor adjustments, the same optical principle could detect trace impurities in water, demonstrating the potential for environmental monitoring.
Thrombosis, or the formation of blood clots in vessels or on implant surfaces, remains a major cause of device failure, making accurate clot measurement critical. The IIT-M team’s method addresses this challenge, offering a reliable tool to improve patient safety and outcomes.
The innovation aligns with India’s policy and legal framework, including the Patents Act, 1970, which protects intellectual property rights, and regulations such as the Medical Devices Rules, 2017, and the National Health Policy, 2017, which emphasize quality, safety, and domestic manufacturing. Key concepts include haemocompatibility (the ability of a material to safely contact blood), thrombosis, turbidity (cloudiness in a fluid), and substrate (the material layer where processes occur).
By replacing mechanical estimation with optical precision, this development marks a step forward in medical technology in India, supporting the indigenization of medical devices and offering potential dual-use applications in environmental monitoring. It also ties to broader initiatives like Make in India and is relevant to science, technology, and health discussions in policy contexts.



