The future of regenerative medicine lies in Anthrobots, miniature biological robots designed for neuron repair and tissue healing, heralding new therapeutic innovations.
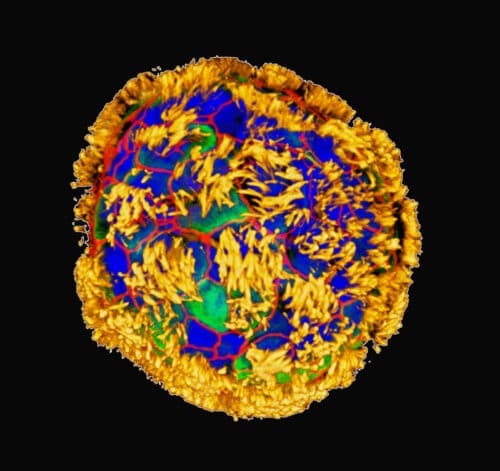
Researchers from Tufts University and the Wyss Institute at Harvard University have developed biologically named Anthrobots using human tracheal cells. These robots can move on surfaces and potentially promote neuron growth across damaged areas in laboratory settings.
How are Anthrobots made?
The researchers refined growth conditions to orient the cilia outward on organoids, marking the first significant observation in their robotics platform. They noted various shapes and movement types, indicating the potential for these Anthrobots to navigate and function within the body or assist in constructing engineered tissues in the lab. The team categorised the Anthrobots, which ranged from 30 to 500 micrometres in size. These bots were either spherical and fully ciliated, irregular or football-shaped with sporadic cilia coverage, or covered on one side. Their movements varied from straight lines and tight circles to combinations of these or simple wiggling. Spherical, cilia-covered bots primarily wiggled, while those with uneven cilia distribution moved more consistently in straight or curved paths. Typically, the Anthrobots lasted 45-60 days in lab conditions before biodegrading naturally.
Little healers
The team, focusing on the therapeutic potential of Anthrobots, designed a lab experiment to evaluate their wound-healing capabilities. They grew a neuron layer from neural stem cells and simulated a wound by scratching it. To increase Anthrobot exposure to the wound, they created “superbots” – clusters of primarily circling and wiggling Anthrobots. Surprisingly, even without genetic modifications, these Anthrobots significantly promoted neural regrowth, bridging gaps as effectively as the surrounding healthy cells, a phenomenon not observed in Anthrobot-free areas. This experiment suggests that Anthrobot assemblies could effectively heal live neural tissue. The team believes these biobots could have broader applications, such as clearing arterial plaque, repairing nerve damage, detecting harmful cells, or delivering targeted drug treatments, potentially aiding in tissue healing and dispensing pro-regenerative drugs.
Making new blueprints, restoring old ones
Utilising the flexibility of cellular assembly, scientists can build biobots and unravel how genomes and environments interact to form and potentially regenerate tissues, organs, and limbs.
Reference: Motile Living Biobots Self-Construct from Adult Human Somatic Progenitor Seed Cells, Advanced Science (2023).



