Read Part 1
Quantum-dot solar cells based on perovskites materials will convert not just rooftops but complete buildings, including windows, into solar energy generators. Up to 26% efficiency has been achieved by mechanically combining perovskite with silicon solar cells.
Quantum dots are already being used in quantum-dot televisions. These have revolutionised the display industry with ultra-high-definition colours and greatly increased effective viewing angles, eliminating the need for rare earth elements—minerals where China has a virtual monopoly in the market.
Quantum dots, however, can also be used to absorb light to boost the output of photovoltaics, photocatalysts, light sensors and other optoelectronic devices. In fact, quantum-dot solar cells can absorb energy 24×7. While conventional silicon modules can absorb visible light during peak times of the day, quantum-dot solar cells can absorb ultraviolet through visible to infrared lighting spectrum to produce power day and night.
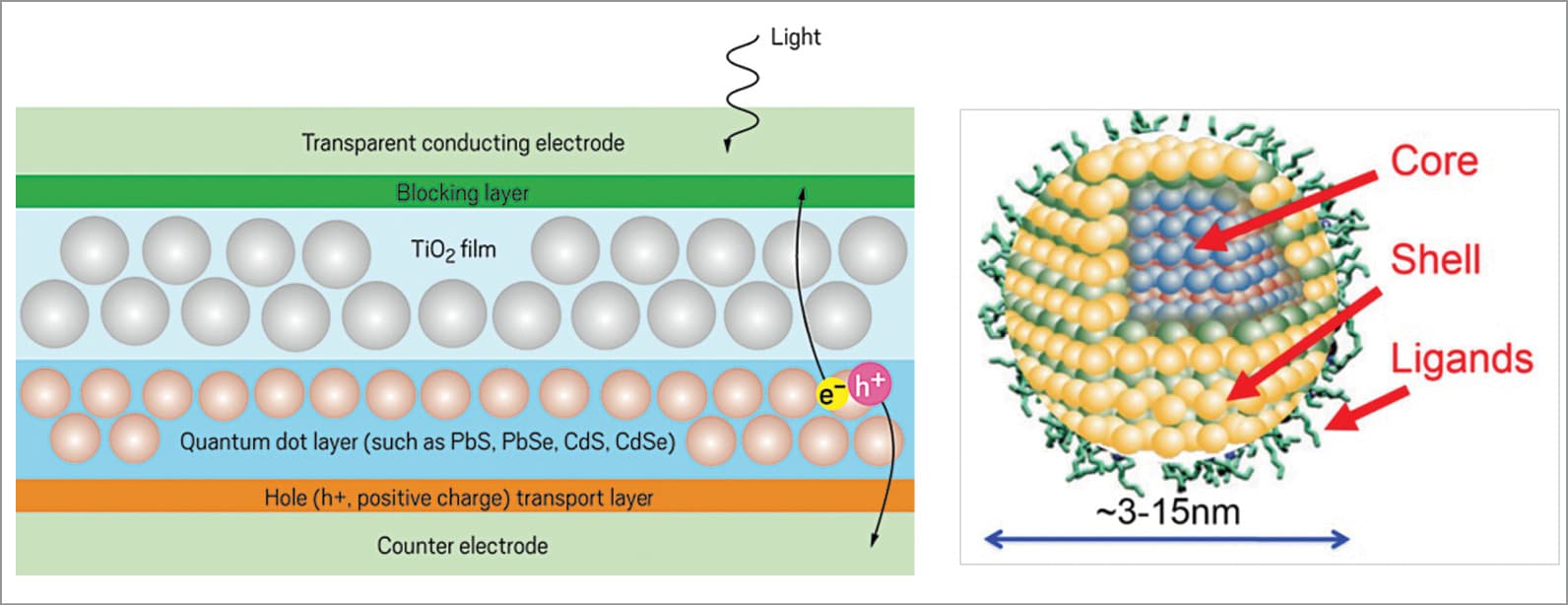
Through a process called multiple exciton generation, quantum dots capture excess photon energy that is normally lost to heat generation. The incident light radiations enter through the transparent electrode of a quantum-dot solar cell onto a light-absorbing layer of dots and create electron-hole pairs (e–/h+). The charged particles then separate and eventually travel to their respective electrodes, thereby producing electric current.
Quantum dots are made from electro-optically active materials, whose size as well as composition controls the photon energy that they interact with, allowing their absorption (and emission) wavelength to be tuned by particle size. These can be tuned to deliver multiple electron-hole pairs from one photon—a process called multiple exciton generation that has allowed quantum-dot solar cells to operate at 10 per cent efficiency.
In quantum-dot solar cells, the function of light-absorbing material is carried out by nanocrystals of semiconducting metal chalcogenides such as CdS, CdSe, PbS and PbSe. Like other emerging photovoltaic technologies, quantum-dot cells can be prepared via low-cost solution-phase chemistry methods and are amenable to high-speed printing techniques.
The structure of quantum dots is peculiar with generally cadmium selenide as the inner core and a Cd1−xZnxS outer shell, coated in silica to avoid oxidation. The outer shell acts like an absorber. When a photon interacts with a quantum dot, the electron transfers from its valence band into the conduction band, leaving a hole in the valence band. The cell is designed such that the shell only absorbs high-energy photons, and the new photon from the core can propagate via internal reflections throughout the glass and quantum-dot layers. Finally, the propagating photons would reach the glass edges, where one or more solar cells could pick them up.
Skyscraper buildings with large windows could harness light for electricity. Researchers at the Los Alamos National Laboratory in New Mexico have reported that a thin film of quantum dots on glass could be the solution to achieving satisfactory efficiency in window photovoltaic systems at a low cost.
A thin layer of quantum dots on normal glass could have a lifetime of up to 14 years but low overall energy conversion efficiency of 1.9 per cent, which should be raised to 6 per cent for practical use. It’s amazingly easy to add quantum dots to window glass as a machine pours slurry of quantum dots and polyvinylpyrrolidone polymer onto the glass and a blade spreads it out to form a thin sheet.
Quantum-dot solar cells may bring breakthrough innovation in the design of solar arrays with their favourable power-to-weight ratio and high efficiency. Mass and area savings as well as flexibility will result in miniaturisation, lower power consumption, increased efficiency, versatility and improved functionality of future satellites. The research also paves the way for new opportunities in the terrestrial renewable energy sector.
Quantum-dot solar window technology converts not just rooftops but complete buildings, including windows, into solar energy generators. It is envisaged that highly efficient and large-area colourless luminescent solar concentrators using heavy-metal-free colloidal quantum dots will soon be available in the market. Luminescent solar concentrators use a new sunlight harvesting technology that has the potential to turn any window into a daytime power source. Many academic research groups have shown that this technology holds promise. But very few companies such as Solterra Renewable Technologies in Texas claim to be competent in this area as it requires unique material chemistry and manufacturing know-how.
Solterra’s cells are designed to exploit Quantum’s low-cost synthesis methods for making four-armed quantum dots. The unique shape of these crystals reduces the probability of electron-hole recombination, which leads to greater charge transport and photovoltaic efficiencies than with spherical quantum dots of the same material.
Quantum-dot solar cells can be made more efficient but further progress in this area has been impeded by the challenge of understanding the mechanisms of electrical conductance in quantum-dot solids and the processes that limit the charge transport distance and low efficiency. The Los Alamos Lab of USA has recently solved this problem. This will lead to a dramatic boost in the photo-voltage and overall device efficiency of quantum-dot solar cells. As economies of scale are achieved, quantum-dot solar cells will reach the market at competitive prices.
Perovskite: The most promising material
Days are not far off when solar cells will be sprayed or printed onto the windows of gigantic buildings, top of cars and walls at an unbelievably low cost. The main driver behind the low price of this technology will be the capability to produce modules as large rolls of a thin film via high-speed processes.
Solar researchers and industries are confident that the miracle perovskites will soon disrupt the economics of over $65-billion industry currently dominated by China. Globally, thousands of researchers are involved in the development of perovskite solar cells. The global efforts and the awesome material properties might finally challenge the 60-year reign of crystalline silicon solar cells.
Historically, the lower price tag of emerging photovoltaics has gone hand in hand with significantly lower performance. If we compare power conversion efficiency of third-generation PVs, dye-sensitised solar cells, organic photovoltaics and quantum-dot cells started at just a few per cent and have climbed slowly over the years to roughly 12 per cent. The value for silicon cells is much higher at 20–25 per cent, but perovskite cells may soon give silicon a run for its money with their efficiency soaring to more than 22 per cent in just a couple of years.
Perovskite solar cells use a perovskite structured compound, generally a hybrid organic-inorganic lead or tin halide-based material with lattice-like structure, which helps in light harvesting. Perovskite materials such as methylammonium lead halides are far inexpensive and relatively simple to manufacture. The unique intrinsic properties of perovskites that make them ideal for solar cell applications are broad absorption spectrum, high absorption coefficient (105cm–1), high dielectric constant, large diffusion length, fast charge separation process, long transport distance of electrons and holes, and long carrier separation lifetime.
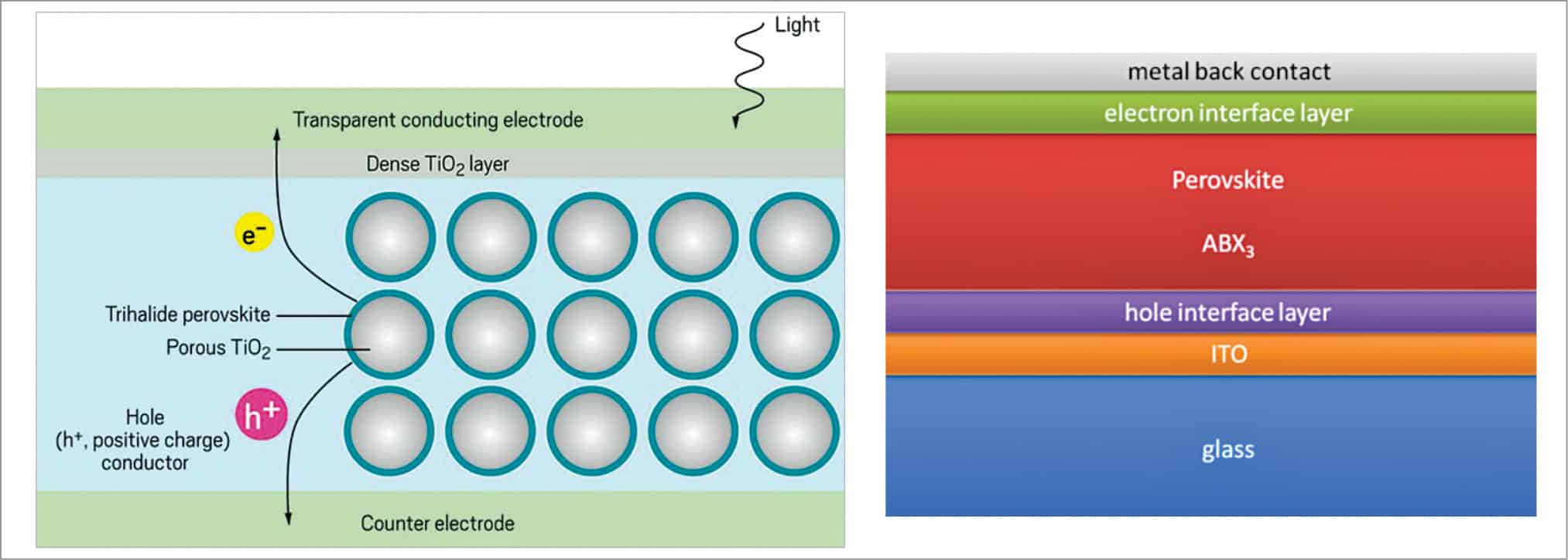
As illustrated in Fig. 8, light incident on the transparent electrode of a perovskite solar cell passes onto a light-absorbing perovskite material layer and creates electron-hole pairs (e–/h+). The charged particles separate due to low binding energy and eventually diffuse through the charge-conducting layers. The charges are then eventually collected by respective electrodes, producing an electric current.
The World Economic Forum declared perovskite as one of the top 10 emerging technologies of 2016. In the meantime, solar panel manufacturers and frontline universities in the US, Europe and Asia are rushing to commercialise this technology.
The perovskite mineral is named after a Russian mineral expert Lev Perovski, who first studied it. Later, researchers found that mineral deposits containing perovskite structures were cheap and abundant throughout the world. Its structure remained poorly understood and ignored for many years as the industry was already biased that silicon was the best material for solar cell applications.
Scientists weren’t sure about perovskite’s usefulness until 2009, when a Japanese researcher Tsutomu Miyasaka, a professor at Toin University of Yokohama, found that perovskite could absorb sunlight and turn it into electricity.
Perovskite presents significant opportunities to realise a low-cost, industry-scalable solar cell technology. This potential for low cost and scalability requires overcoming barriers related to stability and environmental compatibility. If these issues are overcome, a perovskite-based technology could be utilised for terawatt-scale solar deployment.
Scientists are now experimenting with a hybrid structure made of organic molecules and inorganic elements within a single crystalline structure, which together capture light and convert it into electricity. Researchers have achieved 26 per cent efficiency by mechanically combining perovskite with silicon solar cells. These devices use compounds with perovskite crystal structure and stoichiometry to absorb light. (CH3NH3)PbI3 is the most studied example. Scientists do not have clear understanding about their operation mechanism and efficient and fast charge transfer mechanism. Yet, the remarkable rate of progress caused photovoltaics researchers in academia and the industry to switch to perovskites.
Chemicals and materials for perovskite solar-cell R&D are sold by many companies, but none has perovskite modules for sale. Saule Technologies has demonstrated the working prototype of a perovskite module that can power small electronic devices.
Oxford Photovoltaics aims to have a thin-film perovskite solar cell commercially available by year-end. It also plans to design a perovskites-silicon hybrid to produce tandem cells for rooftop panels. As tandem perovskite-silicon cells absorb a larger fraction of the solar spectrum, these are a superior alternative to conventional rooftop silicon panels.
According to report Rise of Perovskites, real commercialisation of perovskite photovoltaics is unlikely to happen until the 2019-21 timeframe. The major obstacle in perovskite commercialisation is stability. The light-sensitive material in perovskite photovoltaic devices dissolves or degrades perovskites within hours in the presence of water vapour and decomposes at high temperature or even by prolonged sun exposure. Methods for painting the material on large surfaces also need improvement.A Stanford University group reported exceptional stability for new perovskite when an organic component called methylammonium was replaced with formamidinium or cesium. When encapsulated to protect against moisture, perovskite cells showed no sign of degradation for six weeks, even when exposed to 85°C temperature and 85 per cent relative humidity—a standard test of durability. Stanford scientists are confident that panels that pass this test will usually not fail due to heat and humidity for over 25 years outside.
Several companies claim to have devised methods for sealing and protecting their cells and modules, but the procedures are proprietary. Scientists must also address the possibility of lead contamination before these cells can be commercialised on a large scale. Proprietary sealing methods may help prevent leakage into the environment. Another strategy under development is replacing lead with metals such as tin.
The inordinate low cost of materials, as well as low manufacturing cost, has led to setting up of commercial perovskite ventures, especially in the United States, aiming for products that could challenge China’s monopoly in the global solar market and help spread manufacturing around the world.
The best is yet to come
The progress of third-generation emerging technologies is unprecedented in photovoltaics history from material development and efficiency advancement perspective. These novel technologies would disrupt the future solar cell market, provided their long-term stability and better efficiency issues are addressed with a better understanding of materials and device operation principles.
Prof. Prashant V. Kamat of University of Notre Dame’s opines that even if the emerging technologies don’t replace silicon cells, these are likely to lead to offshoot technologies and niche applications. At last, we are on the road to see the day when solar becomes an economically substantive part of our energy mix.










