Portable medical devices are improving the healthcare for millions of people. Sophisticated yet affordable, these devices let users monitor their vital signs themselves at home and on the go. Products such as blood glucose monitors, heart rate monitors, ingestible gastrointestinal tract monitors and pain-blocking implants have enhanced the quality of life for those with chronic or acute diseases and conditions.
The coming years will continue to see many new products powered by innovative semiconductor devices.
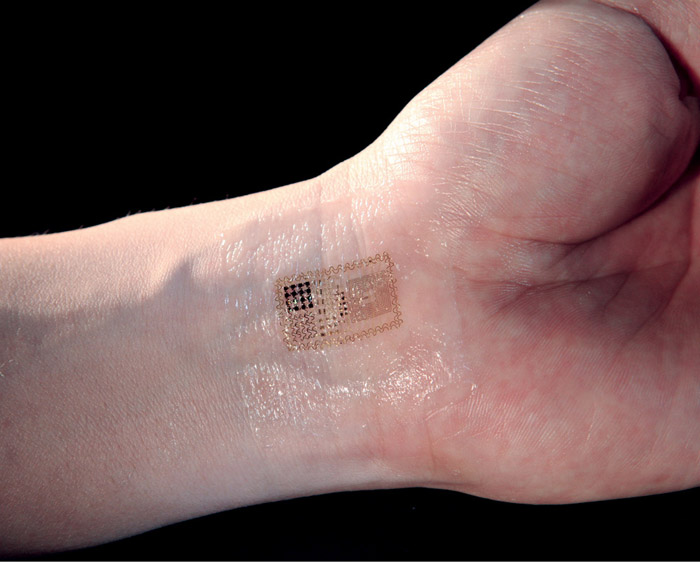
Benefits of going small
While the trend towards miniaturisation is fueled to a large extent by the drive to reduce cost by leveraging the cost advantages generated by Moore’s Law, there are other factors too at play. The most significant among these is the increased focus by semiconductor suppliers on the medical area. Many of them are coming out with more and more solutions tailored to particular applications. The longer-term rationale is that by enabling miniaturisation with more tailored solutions, new markets and users will be created. This will increase volumes and thus financial return on the investment.
A case in point is ultrasound. Traditionally, ultrasound machine was a cart-based unit. More recently, handheld devices have become available with the market no longer limited to hospital use. These devices are little bigger than a mobile phone, packing higher-density measurement electronics such as the AD9278 octal-channel ultrasound receiver from Analog Devices. These miniaturised ultrasound devices have the potential to find their way into the hands of every medical doctor, making ultrasound diagnostics a routine procedure in clinics. One day, they could even replace the stethoscope as the main diagnostic tool of the profession.
Another important driver is the emergence of open mobile phone platform. We are starting to see devices such as glucose meters, heart rate monitors and even pulse oximeters based on mobile phone platforms. The decision to throw open the iPhone and Android platforms has brought about a revolution in medical and healthcare applications. The ultra-low cost and easy availability of mobile phones are compelling arguments in favour of their use in medical field.
One concern, though, is the level of control over such applications. Regulatory bodies such as the Food and Drug Administration (FDA) in the US have been caught off guard by the flood of new devices, and they are now scrambling to address the issue.
Another concern relates to device longevity. The two-year life-cycle of any mobile phone model does not match well with the expected lifecycle of a portable medical device, which could be seven years or more. This raises concerns regarding component supply and product support.
Third is the emergence of low-cost communication standards—both wired and wireless. Larger medical devices are typically used in a hospital or laboratory setting, and results often centralised via computer subsystems connected to a wired Internet connection. The same need for storage of results exists with portable equipment too.
The emergence of wired communication standards such as USB and wireless standards such as Bluetooth have played an important role in easing the communication design and data interoperability challenges. In the wired domain, USB has become ubiquitous, but the wireless situation is still in a state of flux, with Bluetooth (especially Bluetooth Low Energy), Zigbee, the upcoming Medical Body Area Network (MBAN) standard and several others vying for dominance.
Components at the heart
A major trend is the development of devices that can be used by people who have no special training. The component at the centre of this trend is the microcontroller unit (MCU). Making smarter portable devices calls for more capable MCUs. The MCU applies algorithms and programmed data in order to accomplish the intended function of the device.
Besides the MCU, most portable medical-electronic devices have at least one sensor to convert temperature, pressure, sound, electrochemical reactions or some other physical quantity into an electrical signal. These signals often need some level of processing before these can be used by the MCU. This processing is done by analogue components, such as operational amplifiers, filters and analogue-to-digital converters.

MEMS is the technology behind various types of silicon sensors and nano pumps. Consumer sensor technologies from STMicroelectronics include multiple-axis motion and orientation, compass, pressure, temperature, sound (microphone) sensors as well as biological and molecular compounds.
In some portable medical-electronic systems, the MCU needs to be able to affect the outside world. This can be accomplished via motors, linear actuators, pumps, fans, lamps and other components that are connected to the MCU. Depending on the amount of power involved, some of these components may have a field-effect transistor (FET) or other power-control device between them and the MCU.
Most portable medical-electronic devices include some means of communicating with the user. This can be via lights and buttons, an LCD or touch-screen, audio cues including speech, or through a remote interface.
Practically, all of these portable products need a low-power MCU to take commands from the user or operator and provide readings and status updates. Because almost all of these products run on batteries, a key consideration is lengthening the battery life. Products vary in the battery lifetime required, but two to ten years between battery changes is not unusual.
Robust data processing and a wired or wireless communication interface are important. Highly integrated analogue components are required to condition and convert the signals into the digital domain so that these can be processed and interpreted. Portable medical equipment can greatly benefit from the level of integration that the latest SoCs with on-board MCUs provide.
Addressing the key concerns
Semiconductor manufacturers are coming up with innovative solutions to make medical devices portable, low-power, safe, secure, reliable and connected.
Portability. The device should be easy to use wherever the patient happens to be. Here, one very important factor is the physical size of the device, which is partly determined by the size of components used inside.
Semiconductor manufacturers are working hard to reduce both the size and power requirement of portable medical-electronic devices. Consider, for example, accelerometers, which are used in pedometers and blood pressure meters. Devices such as the ADXL345 3-axis accelerometer can operate with a typical power drain of only 25 µA in a 3×5×1mm3 package.
Standby current is especially important in many applications to conserve battery life. Devices such as the ADXL345 can run with standby power levels of 0.1 µA, giving a couple of years of battery life in low-duty-cycle applications.
Power management. It is critical for medical devices to consume minimal power, especially if these are intended to be worn on or implanted inside the human body. The power consumption of medical devices can affect the implant’s lifetime, the amount of heat generated by the device, battery weight and battery cost.
Microcontrollers and other ICs for medical applications achieve low power consumption in a number of ways. Multiple low-power modes, for instance, give designers the flexibility to turn some or all of the MCU functions on or off.
The MCU must support low power in active mode, standby mode and reduced time in an active state. The portable device, and thus the MCU, will be in ‘off’ or lower power state most of the time. However, the device also needs to maintain some sort of function such as clock/calendar or alarm even when not in use. Thus minimising its awake time is the key to extending the battery life. The MCU design should be able to wake the clock and analogue circuits for fast measurements and then allow the MCU to settle back to a low-power state.
A range of ultra-low-power microcontrollers (STM32L and STM8L series from STMicroelectronics) based on a proprietary 130nm ultra-low-leakage process technology are available. These minimise overall energy consumption without compromising on speed and functionality.
In parallel with the drive towards lower current draw, the supply rails required by medical devices are also coming down. While this is relatively easy to achieve in digital circuits as the industry follows the Moore’s Law curve, it is tough for mixed-signal measurement systems. Problems with leakage and signal-to-noise ratio abound. These problems, however, are gradually being brought under control, thus easing the design of the overall power management system.
Connectivity. It is becoming more common for portable medical-electronic devices to communicate with each other or with other devices, either point-to-point or over a network. Connected devices create a number of new capabilities. The simplest may be the one where a device is able to transfer data to a personal computer for storage or analysis. This has so far been done via serial bus or infrared, but latest devices use USB.
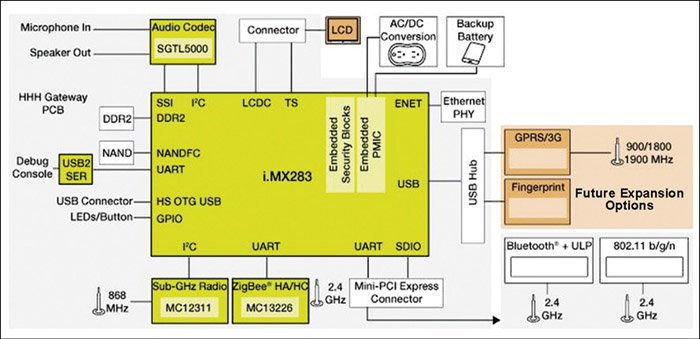
There are also systems that use multiple sensors on a patient to monitor temperature, blood pressure, pulse rate, blood oxygen level, etc. In a networked system, all of these sensors can provide data to a central monitor. Gaining in popularity are systems that gather data from one or more sensors and transfer it to a server via the Internet. This allows patients, or their doctors, to monitor their conditions from anywhere.
Wireless connectivity is redefining the way medicine is practiced. A wireless remote monitoring device can take many forms, but most consist of a wireless sensor connected to a consumer medical device like an ECG, heart monitor or glucometer. These medical end-devices connect wirelessly via the personal area network to an application hosting device, such as mobile phone, PDA, PC, tablet PC or set-top box. This application hosting device then uses a wired (Ethernet) or wireless (cellular) signal to transmit the data to a storage location such as an electronic medical record or personal medical record.
Over time, other more unique form factors that combine multiple consumer medical devices with an application hosting device, like a medical kiosk that is used within hospitals, doctor’s offices and pharmacies, will become prevalent.
Safety. Safety can be enhanced through use of the latest MCUs and sensors. A good example of how technology has improved user safety is in the field of blood-glucose testing for diabetes management.
Years ago, diabetic patients had their blood tested at a doctor’s office every week or so. This provided an incomplete picture of the patient’s glucose level, allowing only coarse control. The advent of portable self-testing glucose meters allows patients to test their blood sugar several times a day, allowing much finer control of glucose levels. At the same time, the transition to electrochemical sensors interfaced to precision analogue components has led to much more accurate readings.
In many ways, miniaturisation actually eases the problem of safety. For example, in many applications, there is a requirement to maintain 5kV isolation between the patient and the main power outlet. As a portable device is often battery-operated, it provides inherent isolation. However, care must still be taken in the system design—for example, when wired communication such as USB is used. Thankfully, help is at hand—semiconductor companies supply USB isolators that comply with the medical standards.
Data security and privacy. The communication link gives an opportunity for the data to be intercepted by an unauthorised party. If the data being transmitted is sensitive medical information, this is clearly unacceptable.
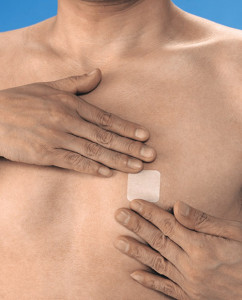
While it is relatively easy to ensure privacy with a wired USB connection, the situation is less robust with wireless connections. Every day, lots of medical information about patients moves through cyberspace, which makes it vulnerable to corruption or interception.
These issues have been comprehensively addressed in standard wireless communication protocols such as Bluetooth. Standards groups such as Continua Alliance have done great work to ensure that data integrity is maintained throughout the complete data chain from portable medical device to hospital and personal health records stored in the cloud.
With increased risk of exposure to threats from cyber-criminals, data encryption is required to establish a secure connection between the provider and the patient.
Data encryption algorithms such as the KEELOQ advanced code-hopping technology can protect data during transmission by rendering it unreadable to unauthorised receivers. When secure encryption is included in the programming of MCUs in distributed data systems, every link in the data chain can be secured.
Experts argue that the most secure connection will be based upon dynamic keying. A direct virtual private network connection will be established based upon continuous rapid sampling of the data stream to change the codec for both the sender and the receiver.
Reliability. Compliance to quality system standards such as ISO/TS-16949 is important for a semiconductor vendor who supplies products to a medical device manufacturer. Microchip’s adherence to the ISO/TS-16949 quality standards aligns well with the requirements of medical device manufacturers for stability and high reliability.
Reliability of the electronics part of portable equipment is generally orders of magnitude higher than other parts of the equipment, such as software algorithms or body contact. Take, for example, fall detectors used for elderly patients. The most common source of a false positive reading is due to algorithmic interpretation rather than raw readings from an accelerometer. Similarly, with ECG measurement systems, an incorrect result is most likely to be caused by poor reliability of the electrode-body contact, rather than the measurement electronics. So it is in these non-electronic areas where most effort must go in order to improve the overall reliability of the device.
Freescale offers comprehensive solutions that help original equipment manufacturers quickly develop ultra-low-power, portable and wireless medical products. Its activity monitor reference design allows recorded data to be interpreted and summarised for improved health performance, enabling consumers to better manage their fitness activities.
Cost. The same technology trends that make MCUs and other electronic components smaller also make them less expensive. Each new generation of semiconductor-fabrication technology allows components to be made smarter at a lower cost. Perhaps, more significant is the fact that as portable medical-electronic devices become more available and commonplace, these enable broader distribution of healthcare to more patients without requiring more doctors. This has a large impact on the overall cost of healthcare.
Dramatic reductions in cost have been achieved by using application-specific semiconductor solutions, as well as low-cost components from high-volume collateral markets such as mobile phones and automobiles.
Latest developments
A very interesting area for the development of new portable medical-electronic devices is drug delivery. We have seen devices that help people take medicines using a variety of methods. These devices contain MCUs and a variety of sensors and actuators to improve the efficiency and accuracy of medicine delivery.
A fascinating example is the electronic drug patch. This patch is affixed to the patient’s skin using some adhesive like a bandage. The material of the patch is covered with a specially formulated drug, which is pushed through the skin into the blood by a controlled electric current. The process is completely painless, and can be self-administered by the patient at home.

Other devices can deliver drugs by injection, inhalation, swallowing or drops in the eyes. In each case, the use of electronics results in more accurate dosing and an automated record to be kept of each dose, resulting in better treatment for the patient.
In addition, many older portable medical-electronic devices are being improved through the addition of friendlier user interfaces. Non-moving touch buttons, proximity detectors and touch screens, which are similar to interfaces on the latest smartphones, are making medical devices easier to use while reducing the possibilities for user error. As these advantages become more broadly known, medical device designers are paying more attention to this type of human-factors engineering.
Freescale Semiconductor recently introduced a home health hub (HHH) reference platform to help medical equipment manufacturers quickly and easily create remote-access devices that can collect, connect and securely share health data for improved healthcare management. The reference platform is based on Freescale’s i.MX28 applications processor and ZigBee and sub-1GHz transceivers. It enables secure Wi-Fi and Ethernet connectivity to remote devices with displays, such as tablets, smartphones or PCs with medical-specific remote user interface options. The platform can also provide wired and wireless connectivity to end-point healthcare devices, such as blood pressure monitors, blood glucometers, weighing scales and pulse oximeters.
Coming soon…
Going forward, it is expected that body-worn sensors will become a very large market. Technical problems remain to be overcome in the sensors required, the algorithms which interpret them and the wireless connectivity required to get the information displayed on an aggregator device such as a mobile phone. These challenges are actively being addressed and we are starting to see emergence of the first devices.
Monica Healthcare, a UK-based company, has launched a disposable adhesive patch device for monitoring pregnant mothers close to delivery. The patch can sense mother and fetus heartbeats as well as abdominal contractions. We can expect to see many more patch devices entering the market.
Also, the introduction of cloud has added a new dimension to the healthcare scenario. 3D printing has been around for a while but now it’s being applied to medicine.
At the recently concluded Consumer Electronics Show in Las Vegas, we saw introduction of a variety of health devices such as Wi-Fi smart scale and sensor patch that if worn for seven days discreetly monitors the activity of the user’s body. This signifies a growing interest in portable medical devices in the consumer segment.
The author is an executive editor at EFY








