Researchers have developed a way to implement neural implants by preventing the formation of scar tissue between the device and the cells.
Scientists around the world have been trying to implement neural implants to restore brain as well as limb function, but have deliberately failed as scar tissue tends to form around the electrodes over time, impeding the connection between the device and the nerve. But a recent study has provided a solution to improve the connection between the neural device and the limb.
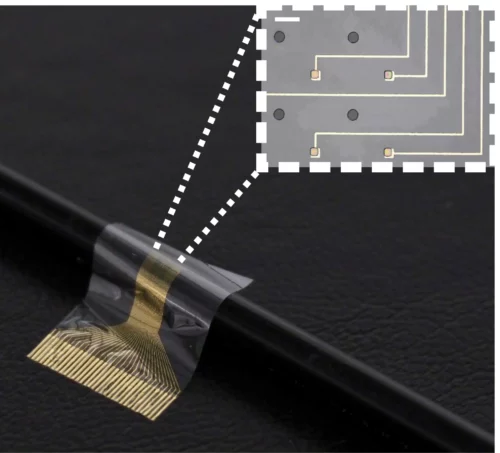
Researchers from the University of Cambridge have developed a novel device that combines flexible electronics and human stem cells – the body’s ‘reprogrammable’ master cells – to better integrate with the nerve and drive limb function. By sandwiching a layer of muscle cells reprogrammed from stem cells between the electrodes and the living tissue, the researchers found that the device integrated with the host’s body and the formation of scar tissue was prevented. The cells survived on the electrode for the duration of the 28-day experiment, the first time this has been monitored over such a long period.
A huge challenge when attempting to reverse injuries that result in the loss of a limb or the loss of function of a limb is the inability of neurons to regenerate and rebuild disrupted neural circuits. One way of addressing this problem is implanting a nerve in the large muscles of the shoulder and attaching electrodes to it. The problem with this approach is scar tissue forms around the electrode, plus it is only possible to extract surface-level information from the electrode. To get better resolution, any implant for restoring function would need to extract much more information from the electrodes. And to improve sensitivity, the researchers wanted to design something that could work on the scale of a single nerve fibre, or axon.
The researchers designed a biocompatible flexible electronic device that is thin enough to be attached to the end of a nerve. A layer of stem cells, reprogrammed into muscle cells, was then placed on the electrode. The Cambridge biohybrid device was implanted into the paralyzed forearm of the rats. While the rats did not have movement restored to their forearms, the device was able to pick up the signals from the brain that control movement. If connected to the rest of the nerve or a prosthetic limb, the device could help restore movement.
Reference : “Functional neurological restoration of amputated peripheral nerve using biohybrid regenerative bioelectronics” by Amy E. Rochford, Alejandro Carnicer-Lombarte, Malak Kawan, Amy Jin, Sam Hilton, Vincenzo F. Curto, Alexandra L. Rutz, Thomas Moreau, Mark R. N. Kotter, George G. Malliaras and Damiano G. Barone, 22 March 2023, Science Advances.
DOI: 10.1126/sciadv.add8162






Just ask if you don’t understand, is this bio-hybrid neural implant also controlled by a chip?
Yes, it’s basically a chip-like device(just like Neuralink’s ‘The Link’ brain implant). However the innovation in this device being discussed above, is in the part that connects the machine/device with the human body. As the device penetrates the skin and makes space for itself inside the body, STEM cells responsible for healing, stick to the device edges and therefore disables proper functioning of the interface.